|
In (b)(i), candidates could not correctly write the three subatomic particles with their corresponding relative masses as they did not understand the demand of the question. In (a)(v), candidates correctly stated that the type of bond which exists in each of ammonia and ammonium ion were covalent bond and covalent bond/dative or coordinate covalent bond respectively. The candidates were also unable to correctly illustrate the formation of ammonium ion with a diagram in (a)(iv). In (a)(iii), most candidates could not correctly show with the aid of a diagram how ammonia molecule is formed. insoluble in water/soluble in non-polar solvents.In (a) (ii), candidates correctly gave two properties of covalent compounds from the following: The expected response from candidates was that covalent bond is a bond between two atoms in which each of the atoms contributes to the shared pair of electrons. In (a)(i), most candidates correctly defined covalent bond however, some of them lost marks because the definition given was not complete. This will take you to the main part of Chemguide.The question was attempted by majority of the candidates and the performance was fair. To return to the list of all the CIE sections To return to the list of learning outcomes in Section 12  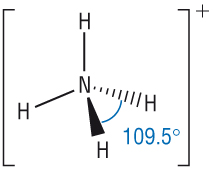
The ammonium salt will react with the base to give off ammonia which will escape into the atmosphere as a gas, and so not be available to the plants. The reason is obvious if you think about it. CIE have on more than one occasion asked why a farmer wouldn't treat a field with an ammonium compound at the same time as using an alkali (a soluble base) like lime. Ammonia gas is produced which (apart from its smell) is recognised because it turns red litmus paper blue.Īmmonium salts such as ammonium sulfate or ammonium nitrate are commonly used as fertilisers. The solid compound (or its solution) is warmed with sodium hydroxide solution. This sort of reaction is also used as a test for ammonium ions in a compound. Ammonium salts react readily with bases to produce ammonia gas.įor example, ammonia is often prepared in the lab by heating an ammonium salt (such as ammonium chloride or ammonium sulfate) with a base such as calcium oxide or calcium hydroxide. Although the electrons are shown differently in the diagram, there is no difference between them in reality.īecause the nitrogen atom has four bonding pairs arranged around it, and no lone pairs, its shape is tetrahedral - just like methane.Īmmonia is only a weak base, and the extra hydrogen ion is easily removed again. Once the ammonium ion has been formed it is impossible to tell any difference between the dative covalent and the ordinary covalent bonds. The hydrogen's electron is left behind on the chlorine to form a negative chloride ion. When the ammonium ion, NH 4 +, is formed, the fourth hydrogen is attached by a dative covalent bond, because only the hydrogen's nucleus is transferred from the chlorine to the nitrogen. Only about 1% has actually produced hydroxide ions.Īmmonia can, of course, react with other more obvious acids to form ammonium salts.įor example, ammonia reacts with hydrogen chloride gas in an acid-base reaction: However, the reaction is reversible, and at any one time about 99% of the ammonia is still present as ammonia molecules. A co-ordinate (dative covalent) bond is formed between the nitrogen and the incoming hydrogen ion. It reacts with water by removing a hydrogen ion from a water molecule to produce ammonium ions and hydroxide ions. You don't need to know about Lewis acids or bases.Īmmonia is a base because of the active lone pair on the nitrogen which can accept hydrogen ions.  
If you aren't familiar with this, you might want to look at the page about theories of acids and bases. It is a Bronsted-Lowry base - a substance which can accept hydrogen ions. This statement deals with the formation and structure of the ammonium ion, and its reactions with bases.īefore you go on, you should find and read the statement in your copy of the syllabus.Īmmonia is a typical weak base. Chemguide: CIE A level chemistry support: Learning outcome 12.1.2Ĭhemguide: Support for CIE A level Chemistry
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
